By Peter A. McCullough, MD, MPH
The use of ivermectin-mebendazole as an adjunct to tradition cancer care is one of the biggest stories coming out of the pandemic. The crisis was destined to have important one or more “spinoff” concepts that make a big impact in medicine. It has been rewarding to see this make the primetime news as Dr McCullough was on set in New York City with Newsmax.
🏥 Potential Breakthrough in Cancer Therapy: Repurposed Medications
In a recent segment on American Agenda, hosts Katrina Szish and Bob Brooks interviewed Dr. Peter McCullough, Chief Scientific Officer of The Wellness Company, regarding a first-of-its-kind human observational report on the off-label use of two generic, repurposed drugs: ivermectin and mebendazole, as an adjunct therapy for cancer patients.
🔬 Key Findings and Mechanisms
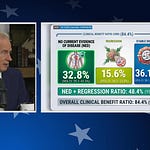
The observational report, currently under peer review, examined patients who chose to integrate these anti-infective medications into their existing cancer treatment regimens to leverage potential off-target anti-cancer properties.
Clinical Outcomes: After six months of use, the report observed that nearly 50% of patients experienced either disease remission or clinical improvement.
Biological Activity: Dr. McCullough highlighted over a decade of preclinical data suggesting these agents:
Reduce cancer cell proliferation.
Inhibit angiogenesis (the blood supply to tumors).
Promote apoptosis (programmed cell death of cancer cells).
Safety Profile: Dr. McCullough emphasized that safety is paramount. While approximately 25% of patients reported side effects, 94% of those were able to continue the treatment, indicating that the combination is generally well-tolerated compared to conventional, high-toxicity cancer therapies.
⚖️ The Call for Institutional Action
Dr. McCullough expressed frustration that repurposed, off-patent medications do not receive the same research investment as proprietary therapies. He argued that the National Cancer Institute has an obligation to explore all treatment avenues, noting that in other medical fields, such as cardiology, the vast majority of drugs utilized are generics.
He advocated for:
Clinical Integration: Inclusion of these drugs in cooperative clinical trial programs.
Rigorous Standardization: Large-scale, prospective, double-blind, randomized, placebo-controlled trials to ascertain efficacy, alongside protocol-driven dose escalation and duration studies.
Joint Decision Making: Until comprehensive trials are completed, the approach remains one of shared decision-making between patients and their physicians.
Please subscribe to FOCAL POINTS as a paying ($5 monthly) or founder member so we can continue to bring you the truth. Alter AI may be used to assist in searches, synthesis, and review.
Peter A. McCullough, MD, MPH
Chief Scientific Officer, The Wellness Company
https://www.twc.health/pages/focal-points
📋 References
Transcript: American Agenda, “Cancer treatment... generic off-patent,” May 6, 2026.
The Wellness Company (twc.health/cancer).