By Peter A. McCullough, MD, MPH
Please enjoy this brief review of ivermectin-mebendazole in cancer treatment as summarized by Dr. McCullough on Real America’s Voice, American Sunrise.
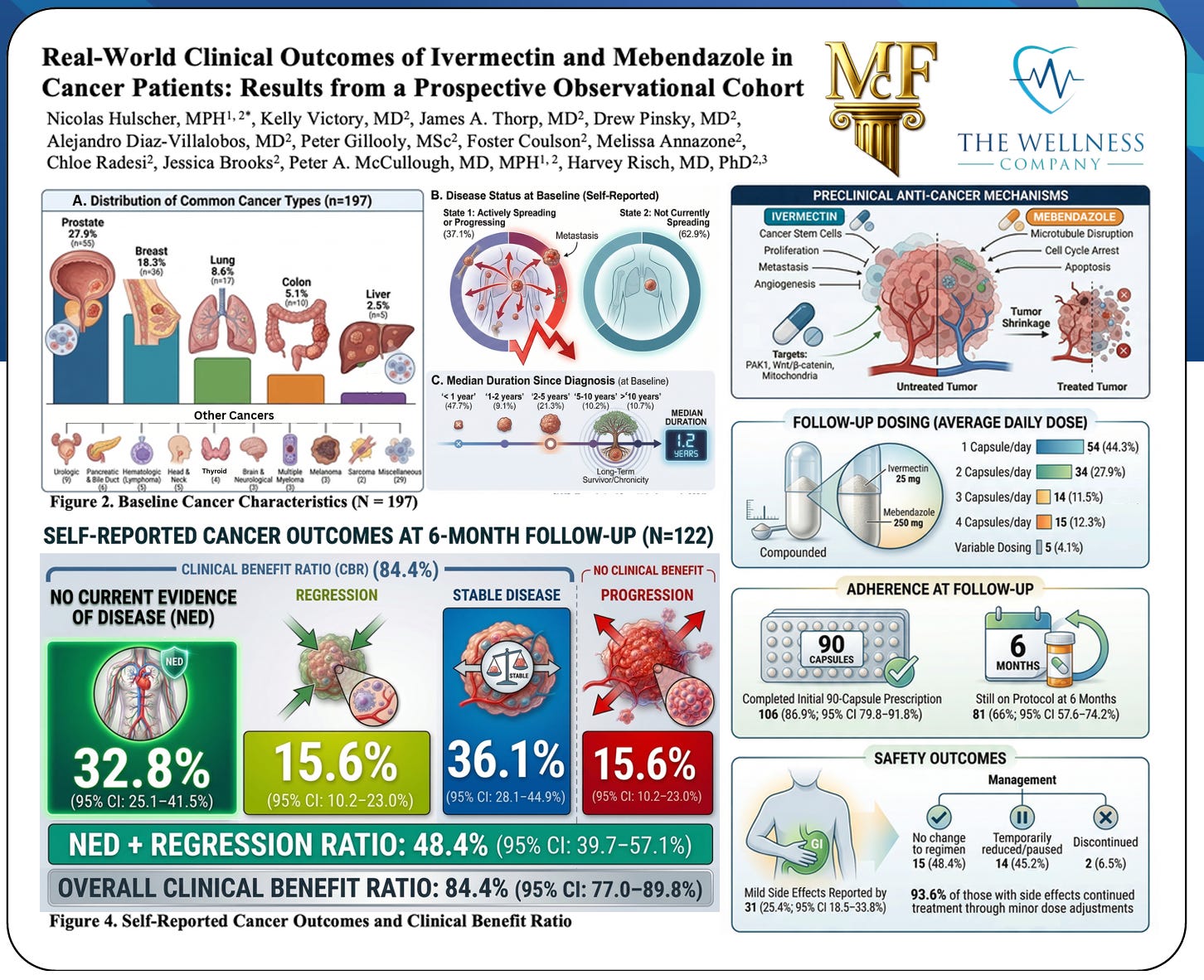
Drug repurposing has emerged as a promising strategy in oncology, and the anti-infective agents ivermectin and mebendazole have attracted attention because of their multi‑target anticancer effects observed in preclinical models. When considered together as adjunctive agents, their complementary mechanisms converge on key hallmarks of cancer: uncontrolled proliferation, angiogenesis, and resistance to apoptosis.
Reduction of cancer cell proliferation is a shared and central effect of both compounds. Mebendazole disrupts microtubule polymerization by binding β‑tubulin, leading to mitotic spindle failure, G2/M arrest, and suppression of rapid cell division. Ivermectin inhibits multiple proliferative signaling pathways, including Wnt/β‑catenin, PAK1, Akt/mTOR, and STAT3, thereby reducing tumor cell growth across diverse cancer models. These mechanisms act independently of classic oncogenic mutations, supporting potential complementary activity. [pmc.ncbi.nlm.nih.gov], [mdpi.com]
Impairment of angiogenesis further constrains tumor expansion. Mebendazole directly suppresses endothelial cell proliferation, migration, and tube formation, even in the presence of pro‑angiogenic growth factors such as VEGF and bFGF. This anti‑angiogenic effect is associated with microtubule disruption and cell‑cycle arrest in endothelial cells. Ivermectin contributes indirectly by downregulating angiogenic signaling and tumor‑supportive pathways, reducing the vascular support required for tumor maintenance. [pmc.ncbi.nlm.nih.gov], [pmc.ncbi.nlm.nih.gov]
Acceleration of apoptosis is a prominent downstream outcome of both agents. Mebendazole induces apoptosis through mitotic catastrophe, caspase‑3 activation, and p53‑associated pathways, while ivermectin promotes programmed cell death via mitochondrial dysfunction, reactive oxygen species generation, and modulation of apoptotic and autophagic signaling. Together, these effects shift cancer cells toward irreversible cell death rather than survival or adaptation. [pmc.ncbi.nlm.nih.gov], [pmc.ncbi.nlm.nih.gov]
Overall, the ivermectin–mebendazole combination illustrates how repurposed drugs may target multiple cancer vulnerabilities simultaneously, and explains why it is biologically plausible that 84% of patients in the recent study from The Wellness Company had net clinical benefit.
Please subscribe to FOCAL POINTS as a paying ($5 monthly) or founder member so we can continue to bring you the truth.
Peter A. McCullough, MD, MPH
Chief Scientific Officer, The Wellness Company
https://www.twc.health/pages/focal-points
References
Tang M. et al. Ivermectin, a potential anticancer drug derived from an antiparasitic drug. Pharmacological Research. 2020. [pmc.ncbi.nlm.nih.gov]
Guerini A.E. et al. Mebendazole as a candidate for drug repurposing in oncology. Cancers (Basel). 2019. [mdpi.com]
Sung S.J. et al. Autophagy is a potential target for enhancing the anti‑angiogenic effect of mebendazole. Biomolecules & Therapeutics. 2019. [pmc.ncbi.nlm.nih.gov]